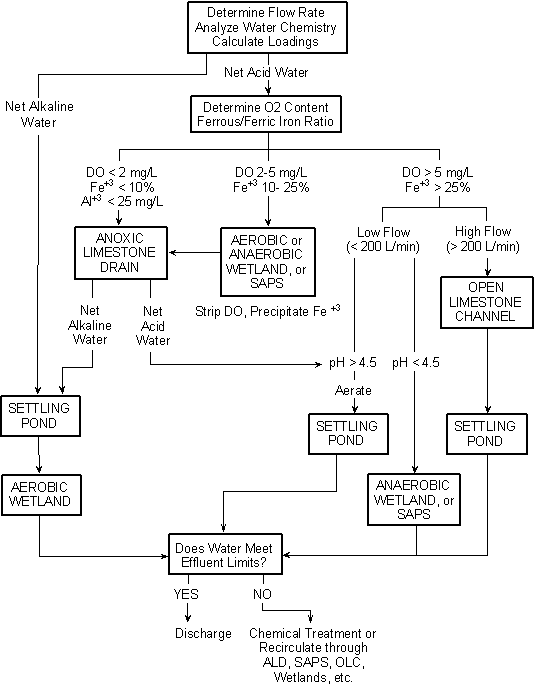
Treatment strategies
Abandoned mine drainage discharges are not all alike. The pollutants, chemical makeup and flow of a given discharge are dependent a variety of factors including the geology and hydrology of the site. Because of this variability between discharge sites, devising a passive treatment strategy will necessarily have to take into account the unique chemical and physical characteristics of the discharge. To better put this situation in perspective, consider the following points.
- In general, treatment of
AMD involves (1) adding alkalinity to the discharge to bring its pH to near
neutral conditions and (2) precipitating and capturing heavy metals present in
the discharge.
- Some AMD discharges are
already net-alkaline (near neutral pH), so the job of adding alkalinity is not
necessary. For these discharges, precipitating and capturing heavy metals
becomes the task.
- In passive treatment
systems, as a general rule, limestone is the favored product used in adding
alkalinity because it is cheap, plentiful, and safe.
- The heavy metals each
exhibit their own behavior in precipitation, both in pH and the physical
characteristics of the precipitate.
- Iron precipitates when it is the ferric oxidation state (iron III) in the form of iron hydroxide Fe(OH)3, yellowboy, as a slimy orange compound at a pH greater than 3.5.
- Aluminum precipitates in the pH greater than 4.5 in the from of aluminum hydroxide Al(OH)3 (gibbsite) as a gelatinous white compound.
- Manganese precipitates
at pH's greater than 8 provided any iron has already precipitated. It is a
black compound.
- When the process of
precipitation of the metal involves the hydrolysis of a metal (as is the case
in the above examples), acidity is added to the water as a byproduct, tending to lower the
pH.
- Fe+3 + 3H2O --> Fe(OH)3 + 3H+
- Al+3 + 3H2O --> Al(OH)3 + 3H+
- Mn+2 + 2H2O
-->
Mn(OH)2 + 2H+
- The first step in the
weathering of pyrite (formation of AMD) places iron in the ferrous (+2) form
that is soluble at all normal pH's.
4FeS2(s) + 14O2(g) + 4H2O(l) ---> 4Fe2+(aq) + 8SO42-(aq) + 8H+(aq) (1)
In other words, as long as iron has not undergone the second reaction (equation 2) whereby the ferrous (+2) iron is oxidized to the ferric (+3) state by reacting with oxygen, no precipitation will occur (as in equation 3).
4Fe2+(aq) + O2(g) + 4H+(aq) ---> 4Fe3+(aq) + 2H2O(l) (2)
Fe3+(aq) + 3 H2O(l) ---> Fe(OH)3(s) + 3H+(aq) (3)The degree to which the second reaction takes place is largely dependent on the amount of dissolved oxygen present in the water and the length of time the ferrous (+2) iron has to react with the oxygen. Some mine discharges have virtually all the iron in the ferrous (+2) state, while others have a large proportion of iron in the ferric (+3) state. However, any proportion of ferrous (+2) to ferric (+3) is possible.
- The use of limestone as an
alkalinity adding agent has limitations. One such limitation occurs when AMD
having sufficient iron in the ferric state passes over the limestone. The
ferric (+3) iron hydrolizes and precipitates as yellowboy (equation 3) coating
the limestone. When enough yellowboy coats the limestone, the limestone
becomes protected from further dissolution and no more alkalinity is added,
thereby defeating the purpose of the limestone. This coating process is known
as armoring and is a failure mode of some treatment systems. If possible
Reaction 2 above needs to be postponed until after the alkalinity is added to
protect limestone from armoring.
- Similar to the armoring process by the precipitation of yellowboy is the precipitation of aluminum hydroxide (gibbsite) with AMD containing significant amount of the metal aluminum. This gelatinous material can accumulate and block the pores of limestone, rendering the limestone useless. Unlike armoring, the gibbsite does not adhere to the limestone, it just blocks it. One treatment system employs a flushing mechanism to wash accumulated gibbsite from the limestone.
In devising a scheme for treating AMD, the characteristics of the discharge play a fundamental role in selecting the combination of treatment strategies to apply.
Settling ponds and constructed aerobic wetlands are a common component of most treatment systems and can be used in combination with other treatment elements. Their purpose is to collect and impound precipitated metals. These are usually the final steps in the treatment process where alkalinity exceeds acidity.
A variety of treatment practices are used to add alkalinity if needed.
Anoxic limestone drains (ALD) are appropriate with discharges that have almost all of the iron in the ferrous state, very little dissolved oxygen, and very little aluminum. Here buried limestone keeps oxygen away from the discharge so that alkalinity can be added, with the chance of armoring minimized.
For more demanding chemistries where a greater proportion of the iron is in the ferric state and more dissolved oxygen is present the use of other treatment practices is warranted.
Successive Alkalinity Producing Systems (SAPS) use a layered approach where mine water filters through a compost layer to remove dissolved oxygen then through a limestone layer to add alkalinity. (The removal of oxygen protects the limestone layer from armoring.) Yellowboy, however, does collect in the system and needs to be flushed periodically.
Anaerobic wetlands (no oxygen) are similar to SAPS in that water passes through organic material and limestone, but without layering. Cattails and other plants are present and play a role in the treatment. Here a different sort of chemistry based on sulfate reduction is employed. Metals are removed and alkalinity added in this system.
The use of Open Limestone Channels has been used successfully in limited circumstances where high flows and a steep slope are possible. The amount of armoring is limited because of the turbulent action of the water and the limestone continues to dissolve effectively.
With these points in mind, it has been possible to create a systematic approach to choosing the treatment regime appropriate to the chemistry and flows as is symbolized in the diagram below.